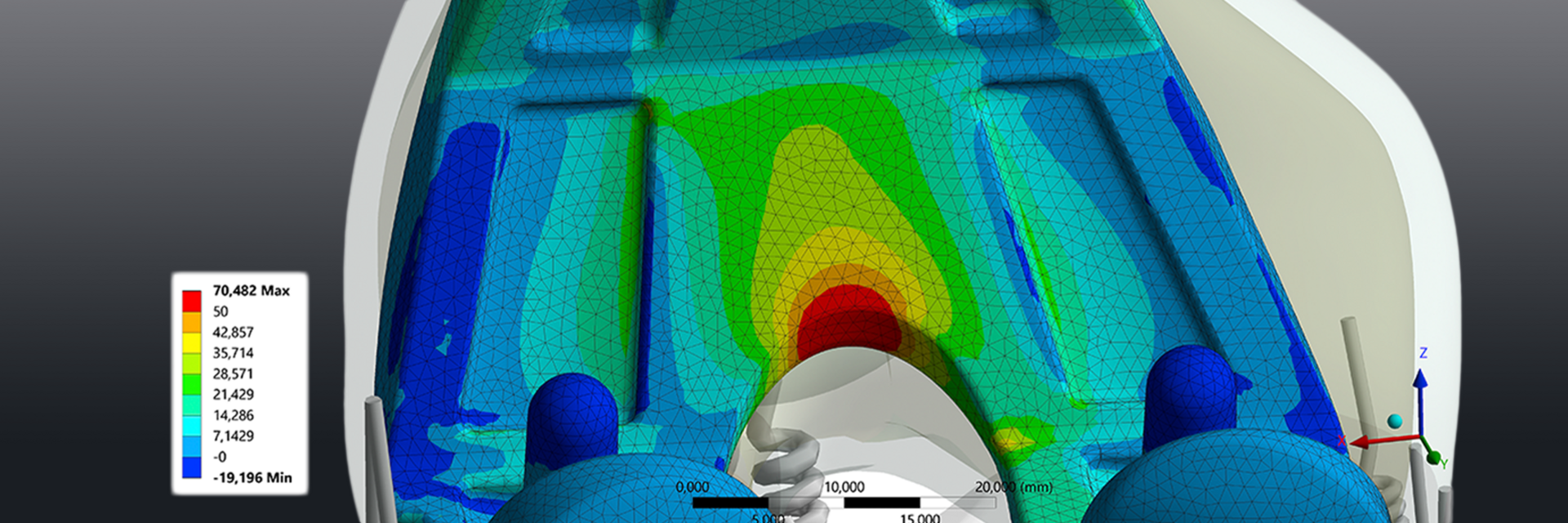
Increase Likelihood of Success
Developing a new product or system is a complex process. Each component needs to be designed while balancing desired functionality and manufacturing costs. The interaction of components influences safety and performance of the system as well.
Frequently, interfaces bear significant failure risk but also present opportunities for superior performance.
In today’s high paced environment, a single R&D department can no longer deal with such complexity. It requires effective and efficient collaboration with other professionals throughout the process.
Over four Decades of Experience
CeramTec has identified the importance of expertise and collaboration across component interfaces and process steps. Therefore, we offer a wide range of engineering services from 3D scanning, CAD modeling and design evaluation through FE-Analyses, risk assessment, and rationales for regulatory purposes, to standard testing, product-specific setups, and statistics.
After a product’s successful market introduction, we offer continuous engineering support, e.g., market surveillance, retrieval analyses, and design changes. CeramTec helps you to achieve superior product performance while meeting your scheduled deadlines.
Engineering at a Glance
Over four decades of experience in designing ceramic components and interfaces for implants
Established development process frequently audited by notified bodies, FDA, and other authorities
Test lab certified according to ISO17025 for a multitude of standardized mechanical tests
Comprehensive database with results of > 5,000 tests and > 3,000 retrieval analyses
Expertise in design and validation of test setups published in peer-reviewed literature
Short communication channels based on our one-stop-shop philosophy