Can ceramic hip resurfacing match the functional outcomes of healthy joints? New peer-reviewed evidence suggests it can.
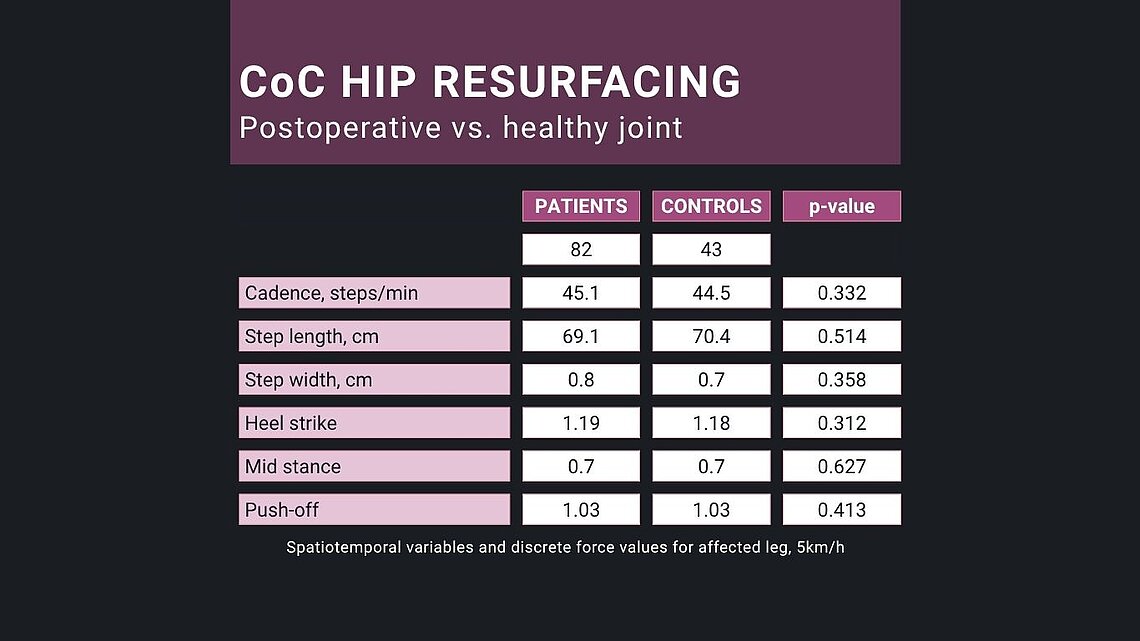
In this prospective, non-randomized clinical study published in @The Bone & Joint Journal, Amy Maslivec and team followed 82 patients (male and female) who received the BIOLOX®delta ceramic-on-ceramic resurfacing H1® (@Embody Orthopaedic/@Zimmer Biomet) and compared them to matched healthy controls.
All procedures were performed by Prof. Cobb. Outcomes were measured using objective gait analysis, Oxford Hip Score (OHS), and Metabolic Equivalent of Task (MET) activity scores.
Key findings
- Pre-op gait abnormalities resolved by 12 months, with gait symmetry index values below 1% at normal walking speeds.
- Hip range of motion (ROM) + walking speed at one year reached levels comparable to healthy controls.
- MET scores improved by 40%, indicating increased post-op activity.
Median OHS rose from 27 to 47.
The authors conclude that the early clinical outcome results of BIOLOX®delta ceramic-on-ceramic resurfacing appear promising, with the potential to restore a near-normal gait pattern and overall quality of life.
These are similar outcomes to metal-on-metal (MoM) hip resurfacing that might include a broader patient population. H1® resurfacing patients achieved top walking speeds and a hip ROM similar to MoM HRA patients, as published in the literature. The difference is that the ceramic design eliminates concerns about metal ion wear and extends eligibility to women and patients with smaller bone sizes who were previously excluded from hip resurfacing procedures.
📖 Maslivec A, Allott N, Guest B, Halewood C, Cobb J. Functional assessment of ceramic-on-ceramic hip resurfacing arthroplasty: improvement in gait at 12 months. Bone Joint J 2026;108-B(4):447–454. doi:10.1302/0301-620X.108B4.
Please check for regulatory approval in your country.
This textt reflects CeramTec’s summary of a peer-reviewed scientific publication and does not constitute clinical guidance, risk prediction for individual patients, or product‑related recommendations. For product, safety, and risk information, always refer to the labeling of the legal manufacturer. This post was drafted with AI assistance and approved by CeramTec.